Institutional Review Board (IRB) Purpose
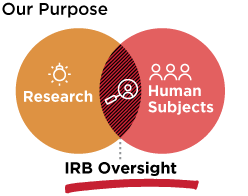
The primary purpose of the IRB is to protect the rights and welfare of human participants involved in research activities. Barry University’s IRB reviews research proposed by faculty, staff, and students. The Office of the Provost oversees the Barry IRB.